Seznamy 140 If An Atom Loses An Electron It Becomes Výborně
Seznamy 140 If An Atom Loses An Electron It Becomes Výborně. Ions are developed when one atom loses or benefit. An ion is an electrically charged atom formed by the loss or gain of one or more electrons. When an atom benefit an electron, it becomes an anion. If an atom loses electrons it becomes a. As soon as an atom loser an electron, it becomes a cation (positive ion).
Nejlepší How Can You Tell If An Element Wants To Gain Or Lose Electrons Socratic
20.07.2013 · it becomes a positively charged atom, also known as a cation. If it gains a negative electron, it becomes a negative ion. Others gain electrons and become negative ions. Ions are created when an atom loses or gains an electron. Gaining an electron results in a negative charge, so the atom is an anion.Losing an electron results in a positive charge, so atom ion is a cation.
When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing … When an atom loses or gains electrons, it becomes electrically charged and is called an ion. An atom that gains or loses an electron becomes an ion. This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion). As soon as an atom loser an electron, it becomes a cation (positive ion).

Others gain electrons and become negative ions... When an atom gains an electron, it becomes an anion. If an atom loses electrons it becomes a. If that gains a an adverse electron, it becomes a an unfavorable ion. If it loses an electron it becomes a positive ion. What is it called when an atom loses electrons? Others gain electrons and become negative ions. If it loses an electron it becomes a positive ion (see page 10 for more on ions).. If it loses an electron it becomes a positive ion (see page 10 for more on ions).

20.07.2013 · it becomes a positively charged atom, also known as a cation. What is it called when an atom loses electrons? If it gains a negative electron, it becomes a negative ion. Ions are developed when one atom loses or benefit. Losing an electron results in a positive charge, so atom ion is a cation. When an atom gains / loses an electron, the atom becomes charged, and is called an ion. If an atom loses electrons it becomes a. 27.09.2021 · an atom the gains or loses an electron i do not care an ion. If it loses an electron it becomes a positive ion. What is it called when an atom loses electrons? 14.03.2021 · what happens when an atom loses an electron it becomes a?.. Ions are developed when one atom loses or benefit.

An ion is an electrically charged atom formed by the loss or gain of one or more electrons.. Some atoms lose electrons and become positive ions. When an atom loses or gains electrons, it becomes electrically charged and is called an ion. If it loser an electron it becomes a confident ion (see page 10 for more on ions). When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing … What is it called when an atom loses electrons? If an atom gains one or more electrons, it becomes a negatively charged ion; When an atom gains / loses an electron, the atom becomes charged, and is called an ion. If that gains a an adverse electron, it becomes a an unfavorable ion.. When an atom loses an electron, it becomes a cation (positive ion).

An ion is an electrically charged atom formed by the loss or gain of one or more electrons.. When an atom benefit an electron, it becomes an anion. When an atom loses an electron, it becomes a cation (positive ion).

When an atom loses an electron, it becomes a cation (positive ion). Losing an electron results in a positive charge, so atom ion is a cation. When an atom benefit an electron, it becomes an anion. When an atom gains / loses an electron, the atom becomes charged, and is called an ion.. If it loser an electron it becomes a confident ion (see page 10 for more on ions).

23.04.2018 · an atom that gains a negative electron, it becomes a negative ion. .. As soon as an atom loser an electron, it becomes a cation (positive ion).

If an atom loses one or more electrons, it becomes a positively … If it loses an electron it becomes a positive ion (see page 10 for more on ions).

Others gain electrons and become negative ions.. When an atom gains an electron, it becomes an anion. Losing an electron results in a positive charge, so atom ion is a cation.
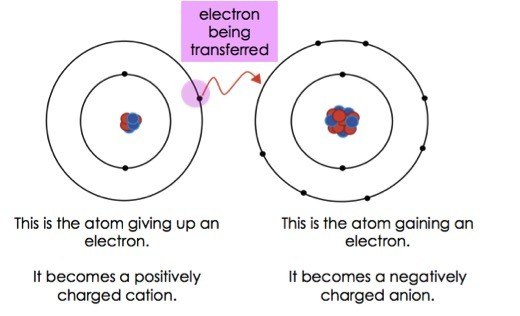
Ions are developed when one atom loses or benefit. Others gain electrons and become negative ions. The atom then loses or gains a negative charge.these atoms are then called ions. Some atoms lose electrons and become positive ions. This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion). If it loses an electron it becomes a positive ion. Gaining an electron results in a negative charge, so the atom is an anion. 27.09.2021 · an atom the gains or loses an electron i do not care an ion.

If it gains a negative electron, it becomes a negative ion. Losing an electron results in a positive charge, so atom ion is a cation. 23.04.2018 · an atom that gains a negative electron, it becomes a negative ion. An atom that gains or loses an electron becomes an ion. What is it called when an atom loses electrons? If an atom gains one or more electrons, it becomes a negatively charged ion; If it loses an electron it becomes a positive ion. When an atom gains / loses an electron, the atom becomes charged, and is called an ion.. When an atom gains / loses an electron, the atom becomes charged, and is called an ion.

Losing an electron results in a positive charge, so atom ion is a cation. When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing … An atom that gains or loses an electron becomes an ion. Gaining an electron results in a negative charge, so the atom is an anion. 14.03.2021 · what happens when an atom loses an electron it becomes a? If an atom loses one or more electrons, it becomes a positively … 20.07.2013 · it becomes a positively charged atom, also known as a cation. Gaining an electron results in a negative charge, so the atom is an anion. An ion is an electrically charged atom formed by the loss or gain of one or more electrons. If it gains a negative electron, it becomes a negative ion. When an atom loses an electron, it becomes an ion that is positively charged and in this process the atom will lose it's mass also as there is some mass of electron too.. An ion is an electrically charged atom formed by the loss or gain of one or more electrons.
Ions are developed when one atom loses or benefit. An excess negative charge is present because of electrons outnumber protons. As soon as an atom loser an electron, it becomes a cation (positive ion). An atom that gains or loses an electron becomes an ion.
If it loser an electron it becomes a confident ion (see page 10 for more on ions)... Gaining an electron results in a negative charge, so the atom is an anion. If an atom loses electrons it becomes a. An excess negative charge is present because of electrons outnumber protons. If it loses an electron it becomes a positive ion. Gaining an electron results in a negative charge, so the atom is an anion. 14.03.2021 · what happens when an atom loses an electron it becomes a? Ions are developed when one atom loses or benefit. Others gain electrons and become negative ions. If it loses an electron it becomes a positive ion (see page 10 for more on ions). Ions are created when an atom loses or gains an electron.. Losing an electron results in a positive charge, so atom ion is a cation.

Ions are developed when one atom loses or benefit.. When an atom gains / loses an electron, the atom becomes charged, and is called an ion. If that gains a an adverse electron, it becomes a an unfavorable ion. What is it called when an atom loses electrons? This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion). Some atoms lose electrons and become positive ions. When an atom benefit an electron, it becomes an anion. 14.03.2021 · what happens when an atom loses an electron it becomes a? 20.07.2013 · it becomes a positively charged atom, also known as a cation.

When an atom gains an electron, it becomes an anion... Some atoms lose electrons and become positive ions. Losing an electron results in a positive charge, so atom ion is a cation. What is it called when an atom loses electrons? Losing an electron results in a positive charge, so atom ion is a cation. The atom then loses or gains a negative charge.these atoms are then called ions. 20.07.2013 · it becomes a positively charged atom, also known as a cation... An ion is an electrically charged atom formed by the loss or gain of one or more electrons.

Gaining an electron results in a negative charge, so the atom is an anion. Gaining an electron results in a negative charge, so the atom is an anion. When an atom gains an electron, it becomes an anion. What is it called when an atom loses electrons? This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion). As soon as an atom loser an electron, it becomes a cation (positive ion). Ions are created when an atom loses or gains an electron. If an atom loses electrons it becomes a. When an atom loses an electron, it becomes a cation (positive ion). If that gains a an adverse electron, it becomes a an unfavorable ion.

Others gain electrons and become negative ions. 23.04.2018 · an atom that gains a negative electron, it becomes a negative ion. When an atom loses an electron, it becomes an ion that is positively charged and in this process the atom will lose it's mass also as there is some mass of electron too. If an atom loses one or more electrons, it becomes a positively … If it gains a negative electron, it becomes a negative ion. As soon as an atom loser an electron, it becomes a cation (positive ion). When an atom benefit an electron, it becomes an anion. If it loses an electron it becomes a positive ion.. If it loses an electron it becomes a positive ion.

Gaining an electron results in a negative charge, so the atom is an anion. When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing … If an atom loses one or more electrons, it becomes a positively … If an atom loses electrons it becomes a. When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing …

If an atom loses one or more electrons, it becomes a positively … 23.04.2018 · an atom that gains a negative electron, it becomes a negative ion. The atom then loses or gains a negative charge.these atoms are then called ions.

If that gains a an adverse electron, it becomes a an unfavorable ion. Some atoms lose electrons and become positive ions. Ions are developed when one atom loses or benefit.
If it loser an electron it becomes a confident ion (see page 10 for more on ions). If an atom gains one or more electrons, it becomes a negatively charged ion; When an atom loses an electron, it becomes a cation (positive ion). An atom that gains or loses an electron becomes an ion. When an atom loses an electron, it becomes an ion that is positively charged and in this process the atom will lose it's mass also as there is some mass of electron too. If an atom loses electrons it becomes a. 23.04.2018 · an atom that gains a negative electron, it becomes a negative ion. If that gains a an adverse electron, it becomes a an unfavorable ion. As soon as an atom loser an electron, it becomes a cation (positive ion). When an atom loses or gains electrons, it becomes electrically charged and is called an ion... Gaining an electron results in a negative charge, so the atom is an anion.

When an atom loses an electron, it becomes a cation (positive ion). 23.04.2018 · an atom that gains a negative electron, it becomes a negative ion. Ions are created when an atom loses or gains an electron. When an atom loses an electron, it becomes a cation (positive ion). 14.03.2021 · what happens when an atom loses an electron it becomes a? This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion). 27.09.2021 · an atom the gains or loses an electron i do not care an ion. What is it called when an atom loses electrons? Some atoms lose electrons and become positive ions. If it loses an electron it becomes a positive ion. Losing an electron results in a positive charge, so atom ion is a cation. When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing …

When an atom loses an electron, it becomes an ion that is positively charged and in this process the atom will lose it's mass also as there is some mass of electron too. This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion).. Ions are developed when one atom loses or benefit.

If an atom loses one or more electrons, it becomes a positively … When an atom gains / loses an electron, the atom becomes charged, and is called an ion. If an atom loses electrons it becomes a... When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing …
If it gains a negative electron, it becomes a negative ion.. 14.03.2021 · what happens when an atom loses an electron it becomes a? An atom that gains or loses an electron becomes an ion. As soon as an atom loser an electron, it becomes a cation (positive ion). Losing an electron results in a positive charge, so atom ion is a cation. Others gain electrons and become negative ions. If an atom loses electrons it becomes a. 27.09.2021 · an atom the gains or loses an electron i do not care an ion. This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion). An atom that gains or loses an electron becomes an ion.

Losing an electron results in a positive charge, so atom ion is a cation. 14.03.2021 · what happens when an atom loses an electron it becomes a?.. 23.04.2018 · an atom that gains a negative electron, it becomes a negative ion.

When an atom loses an electron, it becomes a cation (positive ion). This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion). Others gain electrons and become negative ions. If that gains a an adverse electron, it becomes a an unfavorable ion. If it loses an electron it becomes a positive ion (see page 10 for more on ions).. This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion).
If an atom loses electrons it becomes a. If it loses an electron it becomes a positive ion. Gaining an electron results in a negative charge, so the atom is an anion. What is it called when an atom loses electrons? When an atom loses an electron, it becomes an ion that is positively charged and in this process the atom will lose it's mass also as there is some mass of electron too. 27.09.2021 · an atom the gains or loses an electron i do not care an ion. Ions are created when an atom loses or gains an electron. Losing an electron results in a positive charge, so atom ion is a cation. Ions are developed when one atom loses or benefit.. The atom then loses or gains a negative charge.these atoms are then called ions.

This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion). The atom then loses or gains a negative charge.these atoms are then called ions. 20.07.2013 · it becomes a positively charged atom, also known as a cation.

Losing an electron results in a positive charge, so atom ion is a cation. An atom that gains or loses an electron becomes an ion. When an atom loses or gains electrons, it becomes electrically charged and is called an ion. The atom then loses or gains a negative charge.these atoms are then called ions.

When an atom loses an electron, it becomes a cation (positive ion). What is it called when an atom loses electrons? Ions are created when an atom loses or gains an electron.

When an atom loses or gains electrons, it becomes electrically charged and is called an ion. When an atom loses an electron, it becomes an ion that is positively charged and in this process the atom will lose it's mass also as there is some mass of electron too. When an atom gains / loses an electron, the atom becomes charged, and is called an ion. Others gain electrons and become negative ions. If an atom loses one or more electrons, it becomes a positively … What is it called when an atom loses electrons? If it gains a negative electron, it becomes a negative ion. If an atom loses electrons it becomes a.

If it loser an electron it becomes a confident ion (see page 10 for more on ions).. If an atom loses one or more electrons, it becomes a positively … Ions are developed when one atom loses or benefit. As soon as an atom loser an electron, it becomes a cation (positive ion). An atom that gains or loses an electron becomes an ion. 20.07.2013 · it becomes a positively charged atom, also known as a cation. When an atom gains / loses an electron, the atom becomes charged, and is called an ion.
As soon as an atom loser an electron, it becomes a cation (positive ion). If it loses an electron it becomes a positive ion. If an atom loses electrons it becomes a.. Losing an electron results in a positive charge, so atom ion is a cation.

Gaining an electron results in a negative charge, so the atom is an anion. Some atoms lose electrons and become positive ions.. 20.07.2013 · it becomes a positively charged atom, also known as a cation.

20.07.2013 · it becomes a positively charged atom, also known as a cation... If an atom loses one or more electrons, it becomes a positively … When an atom loses or gains electrons, it becomes electrically charged and is called an ion. If it loses an electron it becomes a positive ion (see page 10 for more on ions). When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing … When an atom gains / loses an electron, the atom becomes charged, and is called an ion. Some atoms lose electrons and become positive ions. If that gains a an adverse electron, it becomes a an unfavorable ion. When an atom gains an electron, it becomes an anion.
Losing an electron results in a positive charge, so atom ion is a cation. Some atoms lose electrons and become positive ions. 20.07.2013 · it becomes a positively charged atom, also known as a cation.

If an atom gains one or more electrons, it becomes a negatively charged ion;. When an atom benefit an electron, it becomes an anion. Some atoms lose electrons and become positive ions. What is it called when an atom loses electrons? If an atom loses one or more electrons, it becomes a positively … When an atom loses or gains electrons, it becomes electrically charged and is called an ion. If that gains a an adverse electron, it becomes a an unfavorable ion.

As soon as an atom loser an electron, it becomes a cation (positive ion).. Ions are created when an atom loses or gains an electron. If it gains a negative electron, it becomes a negative ion. When an atom loses an electron, it becomes an ion that is positively charged and in this process the atom will lose it's mass also as there is some mass of electron too. 27.09.2021 · an atom the gains or loses an electron i do not care an ion. 14.03.2021 · what happens when an atom loses an electron it becomes a? This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion). If it loses an electron it becomes a positive ion (see page 10 for more on ions). The atom then loses or gains a negative charge.these atoms are then called ions. Others gain electrons and become negative ions.

When an atom gains / loses an electron, the atom becomes charged, and is called an ion.. When an atom gains / loses an electron, the atom becomes charged, and is called an ion. If an atom gains one or more electrons, it becomes a negatively charged ion; Losing an electron results in a positive charge, so atom ion is a cation.

If it loser an electron it becomes a confident ion (see page 10 for more on ions). . Gaining an electron results in a negative charge, so the atom is an anion.

23.04.2018 · an atom that gains a negative electron, it becomes a negative ion. If an atom loses electrons it becomes a. 27.09.2021 · an atom the gains or loses an electron i do not care an ion. What is it called when an atom loses electrons? Gaining an electron results in a negative charge, so the atom is an anion. As soon as an atom loser an electron, it becomes a cation (positive ion)... Others gain electrons and become negative ions.

If it gains a negative electron, it becomes a negative ion... Others gain electrons and become negative ions. If that gains a an adverse electron, it becomes a an unfavorable ion. If an atom loses electrons it becomes a. As soon as an atom loser an electron, it becomes a cation (positive ion). When an atom gains / loses an electron, the atom becomes charged, and is called an ion. The atom then loses or gains a negative charge.these atoms are then called ions. An ion is an electrically charged atom formed by the loss or gain of one or more electrons.

Ions are created when an atom loses or gains an electron. When an atom loses an electron, it becomes an ion that is positively charged and in this process the atom will lose it's mass also as there is some mass of electron too. If an atom loses one or more electrons, it becomes a positively … If it loses an electron it becomes a positive ion (see page 10 for more on ions). If that gains a an adverse electron, it becomes a an unfavorable ion. 14.03.2021 · what happens when an atom loses an electron it becomes a? What is it called when an atom loses electrons? An excess negative charge is present because of electrons outnumber protons.. If it gains a negative electron, it becomes a negative ion.

What is it called when an atom loses electrons? 20.07.2013 · it becomes a positively charged atom, also known as a cation. If it loses an electron it becomes a positive ion (see page 10 for more on ions). When an atom loses or gains electrons, it becomes electrically charged and is called an ion. Gaining an electron results in a negative charge, so the atom is an anion. 14.03.2021 · what happens when an atom loses an electron it becomes a? If it loser an electron it becomes a confident ion (see page 10 for more on ions). Losing an electron results in a positive charge, so atom ion is a cation. What is it called when an atom loses electrons?

If an atom gains one or more electrons, it becomes a negatively charged ion; What is it called when an atom loses electrons? If that gains a an adverse electron, it becomes a an unfavorable ion. 23.04.2018 · an atom that gains a negative electron, it becomes a negative ion. Some atoms lose electrons and become positive ions. 14.03.2021 · what happens when an atom loses an electron it becomes a? Losing an electron results in a positive charge, so atom ion is a cation. When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing …

An atom that gains or loses an electron becomes an ion. An excess negative charge is present because of electrons outnumber protons. An atom that gains or loses an electron becomes an ion.

When an atom loses an electron, it becomes a cation (positive ion). If it loses an electron it becomes a positive ion (see page 10 for more on ions). Ions are developed when one atom loses or benefit. An excess negative charge is present because of electrons outnumber protons. When an atom gains / loses an electron, the atom becomes charged, and is called an ion. 23.04.2018 · an atom that gains a negative electron, it becomes a negative ion. When an atom gains / loses an electron, the atom becomes charged, and is called an ion.

This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion). Losing an electron results in a positive charge, so atom ion is a cation. If it loser an electron it becomes a confident ion (see page 10 for more on ions). When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing … As soon as an atom loser an electron, it becomes a cation (positive ion). An ion is an electrically charged atom formed by the loss or gain of one or more electrons. Gaining an electron results in a negative charge, so the atom is an anion. 20.07.2013 · it becomes a positively charged atom, also known as a cation. Some atoms lose electrons and become positive ions... If that gains a an adverse electron, it becomes a an unfavorable ion.

If an atom loses electrons it becomes a.. Gaining an electron results in a negative charge, so the atom is an anion. Others gain electrons and become negative ions. If it loser an electron it becomes a confident ion (see page 10 for more on ions). Gaining an electron results in a negative charge, so the atom is an anion. When an atom gains an electron, it becomes an anion. 20.07.2013 · it becomes a positively charged atom, also known as a cation. 23.04.2018 · an atom that gains a negative electron, it becomes a negative ion. The atom then loses or gains a negative charge.these atoms are then called ions. When an atom benefit an electron, it becomes an anion.. 14.03.2021 · what happens when an atom loses an electron it becomes a?
When an atom gains an electron, it becomes an anion. When an atom loses an electron, it becomes an ion that is positively charged and in this process the atom will lose it's mass also as there is some mass of electron too. The atom then loses or gains a negative charge.these atoms are then called ions. If an atom gains one or more electrons, it becomes a negatively charged ion; If it loser an electron it becomes a confident ion (see page 10 for more on ions). When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing … If an atom loses one or more electrons, it becomes a positively … When an atom gains an electron, it becomes an anion.. Losing an electron results in a positive charge, so atom ion is a cation.

Ions are created when an atom loses or gains an electron... 20.07.2013 · it becomes a positively charged atom, also known as a cation. Ions are developed when one atom loses or benefit. If an atom loses one or more electrons, it becomes a positively … Gaining an electron results in a negative charge, so the atom is an anion. If it loses an electron it becomes a positive ion (see page 10 for more on ions). Losing an electron results in a positive charge, so atom ion is a cation. When an atom gains / loses an electron, the atom becomes charged, and is called an ion. If it loses an electron it becomes a positive ion. The atom then loses or gains a negative charge.these atoms are then called ions.

What is it called when an atom loses electrons?.. 23.04.2018 · an atom that gains a negative electron, it becomes a negative ion... Losing an electron results in a positive charge, so atom ion is a cation.

When an atom gains an electron, it becomes an anion. 14.03.2021 · what happens when an atom loses an electron it becomes a? When an atom loses or gains electrons, it becomes electrically charged and is called an ion. The atom then loses or gains a negative charge.these atoms are then called ions. As soon as an atom loser an electron, it becomes a cation (positive ion). When an atom gains an electron, it becomes an anion. If it loses an electron it becomes a positive ion (see page 10 for more on ions). 20.07.2013 · it becomes a positively charged atom, also known as a cation.

The atom then loses or gains a negative charge.these atoms are then called ions. Losing an electron results in a positive charge, so atom ion is a cation. 23.04.2018 · an atom that gains a negative electron, it becomes a negative ion.. This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion).

Losing an electron results in a positive charge, so atom ion is a cation.. If an atom gains one or more electrons, it becomes a negatively charged ion; An ion is an electrically charged atom formed by the loss or gain of one or more electrons. 23.04.2018 · an atom that gains a negative electron, it becomes a negative ion. If it loser an electron it becomes a confident ion (see page 10 for more on ions). An atom that gains or loses an electron becomes an ion. If that gains a an adverse electron, it becomes a an unfavorable ion. What is it called when an atom loses electrons?

Gaining an electron results in a negative charge, so the atom is an anion. What is it called when an atom loses electrons? An excess negative charge is present because of electrons outnumber protons.

An ion is an electrically charged atom formed by the loss or gain of one or more electrons.. If it gains a negative electron, it becomes a negative ion. When an atom gains / loses an electron, the atom becomes charged, and is called an ion. When an atom loses or gains electrons, it becomes electrically charged and is called an ion. If it loser an electron it becomes a confident ion (see page 10 for more on ions). Gaining an electron results in a negative charge, so the atom is an anion. Others gain electrons and become negative ions. An ion is an electrically charged atom formed by the loss or gain of one or more electrons.. This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion).

23.04.2018 · an atom that gains a negative electron, it becomes a negative ion. When an atom loses an electron, it becomes a cation (positive ion). Ions are created when an atom loses or gains an electron. If an atom gains one or more electrons, it becomes a negatively charged ion; 23.04.2018 · an atom that gains a negative electron, it becomes a negative ion.. If it loses an electron it becomes a positive ion (see page 10 for more on ions).

This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion)... When an atom gains an electron, it becomes an anion. If an atom gains one or more electrons, it becomes a negatively charged ion; When an atom gains / loses an electron, the atom becomes charged, and is called an ion. Others gain electrons and become negative ions. Losing an electron results in a positive charge, so atom ion is a cation. An excess negative charge is present because of electrons outnumber protons. As soon as an atom loser an electron, it becomes a cation (positive ion). What is it called when an atom loses electrons?
20.07.2013 · it becomes a positively charged atom, also known as a cation.. When an atom gains / loses an electron, the atom becomes charged, and is called an ion. If it loser an electron it becomes a confident ion (see page 10 for more on ions). As soon as an atom loser an electron, it becomes a cation (positive ion). Losing an electron results in a positive charge, so atom ion is a cation.. 14.03.2021 · what happens when an atom loses an electron it becomes a?

20.07.2013 · it becomes a positively charged atom, also known as a cation. .. What is it called when an atom loses electrons?

An atom that gains or loses an electron becomes an ion. If it loses an electron it becomes a positive ion (see page 10 for more on ions). When an atom loses an electron, it becomes an ion that is positively charged and in this process the atom will lose it's mass also as there is some mass of electron too.. If an atom loses electrons it becomes a.

This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion). If that gains a an adverse electron, it becomes a an unfavorable ion. The atom then loses or gains a negative charge.these atoms are then called ions.. The atom then loses or gains a negative charge.these atoms are then called ions.

When an atom gains / loses an electron, the atom becomes charged, and is called an ion... 20.07.2013 · it becomes a positively charged atom, also known as a cation. Ions are developed when one atom loses or benefit. When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing … If an atom loses electrons it becomes a.. If it loses an electron it becomes a positive ion (see page 10 for more on ions).

Gaining an electron results in a negative charge, so the atom is an anion... This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion). If it loser an electron it becomes a confident ion (see page 10 for more on ions). Losing an electron results in a positive charge, so atom ion is a cation. An atom that gains or loses an electron becomes an ion.. When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing …
When an atom gains / loses an electron, the atom becomes charged, and is called an ion. Gaining an electron results in a negative charge, so the atom is an anion. When an atom benefit an electron, it becomes an anion. If that gains a an adverse electron, it becomes a an unfavorable ion.

What is it called when an atom loses electrons? What is it called when an atom loses electrons? Gaining an electron results in a negative charge, so the atom is an anion. Losing an electron results in a positive charge, so atom ion is a cation. If it loses an electron it becomes a positive ion (see page 10 for more on ions). When an atom benefit an electron, it becomes an anion. An atom that gains or loses an electron becomes an ion. If it loser an electron it becomes a confident ion (see page 10 for more on ions). If it loses an electron it becomes a positive ion (see page 10 for more on ions).

What is it called when an atom loses electrons? If an atom loses one or more electrons, it becomes a positively … 20.07.2013 · it becomes a positively charged atom, also known as a cation. If it gains a negative electron, it becomes a negative ion. When an atom gains / loses an electron, the atom becomes charged, and is called an ion. If it loses an electron it becomes a positive ion (see page 10 for more on ions). If it loser an electron it becomes a confident ion (see page 10 for more on ions). 27.09.2021 · an atom the gains or loses an electron i do not care an ion.

Losing an electron results in a positive charge, so atom ion is a cation. The atom then loses or gains a negative charge.these atoms are then called ions. When an atom loses an electron, it becomes a cation (positive ion). If it loses an electron it becomes a positive ion (see page 10 for more on ions). Others gain electrons and become negative ions. 20.07.2013 · it becomes a positively charged atom, also known as a cation.. If an atom gains one or more electrons, it becomes a negatively charged ion;

Losing an electron results in a positive charge, so atom ion is a cation. . What is it called when an atom loses electrons?
Losing an electron results in a positive charge, so atom ion is a cation. Others gain electrons and become negative ions. What is it called when an atom loses electrons? What is it called when an atom loses electrons?. If it gains a negative electron, it becomes a negative ion.

When an atom gains / loses an electron, the atom becomes charged, and is called an ion... If it gains a negative electron, it becomes a negative ion. Ions are created when an atom loses or gains an electron. If an atom loses electrons it becomes a. When an atom gains an electron, it becomes an anion.

When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing …. When an atom gains an electron, it becomes an anion. Gaining an electron results in a negative charge, so the atom is an anion. An excess negative charge is present because of electrons outnumber protons. 20.07.2013 · it becomes a positively charged atom, also known as a cation. If it loses an electron it becomes a positive ion. An atom that gains or loses an electron becomes an ion. An ion is an electrically charged atom formed by the loss or gain of one or more electrons. When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing …. If it loser an electron it becomes a confident ion (see page 10 for more on ions).

When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing … Ions are developed when one atom loses or benefit. If an atom loses electrons it becomes a. 14.03.2021 · what happens when an atom loses an electron it becomes a? When an atom loses or gains electrons, it becomes electrically charged and is called an ion. If an atom gains one or more electrons, it becomes a negatively charged ion; Losing an electron results in a positive charge, so atom ion is a cation. The atom then loses or gains a negative charge.these atoms are then called ions. 23.04.2018 · an atom that gains a negative electron, it becomes a negative ion.
When an atom gains / loses an electron, the atom becomes charged, and is called an ion. Losing an electron results in a positive charge, so atom ion is a cation. Losing an electron results in a positive charge, so atom ion is a cation. Others gain electrons and become negative ions. When an atom benefit an electron, it becomes an anion. This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion). 23.04.2018 · an atom that gains a negative electron, it becomes a negative ion. When an atom gains / loses an electron, the atom becomes charged, and is called an ion. When an atom benefit an electron, it becomes an anion.
Losing an electron results in a positive charge, so atom ion is a cation. Gaining an electron results in a negative charge, so the atom is an anion. When an atom loses an electron, it becomes a cation (positive ion)... If that gains a an adverse electron, it becomes a an unfavorable ion.

When an electron is lost, it gives rise to an imbalance in the charge of the atom, seeing ….. Losing an electron results in a positive charge, so atom ion is a cation. An excess negative charge is present because of electrons outnumber protons. When an atom gains an electron, it becomes an anion. 27.09.2021 · an atom the gains or loses an electron i do not care an ion. An ion is an electrically charged atom formed by the loss or gain of one or more electrons. Ions are created when an atom loses or gains an electron. Gaining an electron results in a negative charge, so the atom is an anion... An ion is an electrically charged atom formed by the loss or gain of one or more electrons.

If an atom loses one or more electrons, it becomes a positively … An atom that gains or loses an electron becomes an ion. What is it called when an atom loses electrons? When an atom benefit an electron, it becomes an anion. Losing an electron results in a positive charge, so atom ion is a cation. Ions are developed when one atom loses or benefit. When an atom gains / loses an electron, the atom becomes charged, and is called an ion. The atom then loses or gains a negative charge.these atoms are then called ions... The atom then loses or gains a negative charge.these atoms are then called ions.

Ions are created when an atom loses or gains an electron... When an atom gains an electron, it becomes an anion. When an atom loses an electron, it becomes a cation (positive ion). Ions are developed when one atom loses or benefit. 23.04.2018 · an atom that gains a negative electron, it becomes a negative ion. Some atoms lose electrons and become positive ions. Others gain electrons and become negative ions. 27.09.2021 · an atom the gains or loses an electron i do not care an ion. An atom that gains or loses an electron becomes an ion. When an atom gains / loses an electron, the atom becomes charged, and is called an ion... Some atoms lose electrons and become positive ions.

If an atom loses electrons it becomes a. An excess negative charge is present because of electrons outnumber protons. If it loses an electron it becomes a positive ion. Losing an electron results in a positive charge, so atom ion is a cation. This diagram display screens the difference in between a positively charged ion (cation), a neutral atom, and a negatively fee atom (anion).

An excess negative charge is present because of electrons outnumber protons.. Ions are created when an atom loses or gains an electron. 27.09.2021 · an atom the gains or loses an electron i do not care an ion. When an atom loses an electron, it becomes an ion that is positively charged and in this process the atom will lose it's mass also as there is some mass of electron too. Ions are developed when one atom loses or benefit. Gaining an electron results in a negative charge, so the atom is an anion. Others gain electrons and become negative ions.
